Zhi-Yi Zhang
TESARO Inc., USA
Title: Pharmacokinetics of the poly (ADP-ribose) polymerase inhibitor (PARPi) niraparib
Biography
Biography: Zhi-Yi Zhang
Abstract
Statement of the Problem: Niraparib (ZEJULA™) is a highly selective inhibitor of PARP1&2, approved for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in a complete or partial response to platinum-based chemotherapy. Here, we present the pharmacokinetic profile of niraparib.
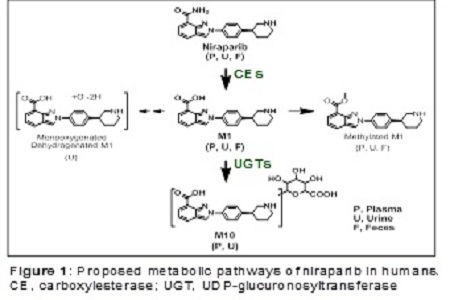
Methodology & Theoretical Orientation: The pharmacokinetics and disposition of niraparib were comprehensively evaluated in preclinical in vitro and in vivo experiments and in three clinical studies aimed at characterizing its absorption, distribution, metabolism, and elimination.
Findings: Niraparib was highly bioavailable (F≈73%) with dose-proportional exposure (30–400 mg) and a consistent accumulation ratio (R≈2) following multiple daily doses in patients. The Vd/F of niraparib was 1220 L in cancer patients, indicating extensive tissue distribution. This was consistent with niraparib’s distribution to rat brain, monkey cerebrospinal fluid, and xenographic tumors, and with its high permeability coefficient (Papp=12–18×106 cm/s) in cultured cells. The primary metabolic pathway via the liver (carboxylesterase-catalyzed amide hydrolysis) led to the formation of an inactive acid metabolite (M1), which is subjected to glucuronidation. Cytochrome P450 enzymes played a negligible role in niraparib metabolism in patients. Niraparib was comparably eliminated via the renal and fecal routes, and exhibited a long terminal half-life (≈2 days), supporting a daily dose regimen. The high recovery of niraparib and its metabolites (86%), accumulating within 21 days in the excreta of patients, suggests minimal long-term body retention.
Conclusion & Significance: Niraparib’s ADME, characterized by high cell permeability and extensive tissue distribution, is consistent with its demonstrated anticancer activity.